VO: In this video, you will see the novel mechanism of action for first-in-class RUKOBIA, the first Food and Drug Administration-, or FDA-, approved attachment inhibitor for HIV-1.
VIDEO: RUKOBIA logo and branding colors, there will be slight movement for all the elements in the scene.
VO: Indication. RUKOBIA, used with other antiretrovirals, is indicated for heavily treatment-experienced adults with multidrug-resistant HIV-1 who are failing their current ARV regimen.
Important Safety Information. Contraindications. Do not use in patients with previous hypersensitivity to fostemsavir or any of the components of RUKOBIA. Do not use RUKOBIA in patients receiving strong cytochrome P450 (CYP)3A inducers, including those listed here. Please see additional Important Safety Information for RUKOBIA at the end of the video. Please click the link on this page to view the full Prescribing Information for RUKOBIA.
VIDEO: Text softly fades into the background, Frame 3 fades in from the center.
VO: The outer gp120 protein mediates initial attachment to the CD4+ T-cell.
SUPERS: gp41; gp120; surface glycoprotein; HIV-1 viral membrane.
VIDEO: Surface of HIV-1 and trimer.
VO: The gp120/gp41 trimer dynamically transitions between open and closed states.
SUPERS: Open state, HIV-1 viral membrane.
VIDEO: Surface of HIV-1 and open trimer. Staying on the close shot of the trimer, supers from Frame 3 fade out and new super fades in.
VO: When in the open state, the gp120/gp41 trimer is able to bind to the CD4+ T-cell.
SUPERS: HIV-1 viral membrane.
VIDEO: Surface of HIV-1 and open trimer. Staying on the close shot of the trimer, super from Frame 4 fades out and new super fades in.
VO: The three main steps of viral entry are gp120-CD4 attachment, co-receptor binding, and membrane fusion. Attachment occurs when the gp120/gp41 trimer is in the open state and binds to the CD4 receptor, causing a series of structural changes in the gp120/gp41 trimer. This allows the subsequent binding of gp120 to a cellular surface co-receptor, either CCR5 or CXCR4.
SUPERS: Binding of co-receptors; CD4+ T-cell; CD4+ T-cell attachment receptor; CCR5.
VIDEO: The trimer in its open state animates to demonstrate co-receptor binding of gp120 with CD4+ T-cell receptor, followed by binding of surface co-receptor CCR5/CXCR4. Super from Frame 5 fades out and new supers fade in.
VO: This sequential binding allows for further rearrangements in the glycoprotein complex, which drives the fusion of viral and cellular membranes.
SUPERS: N/A
VIDEO: Animation continues to show co-receptor binding and cell membrane fusion. Supers from Frame 6 fade out.
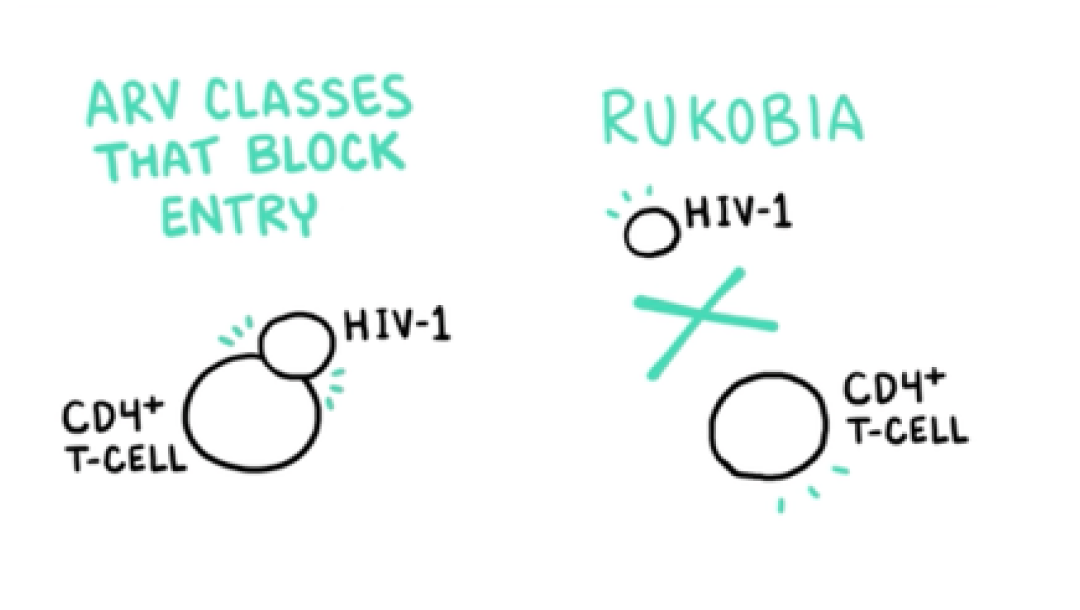
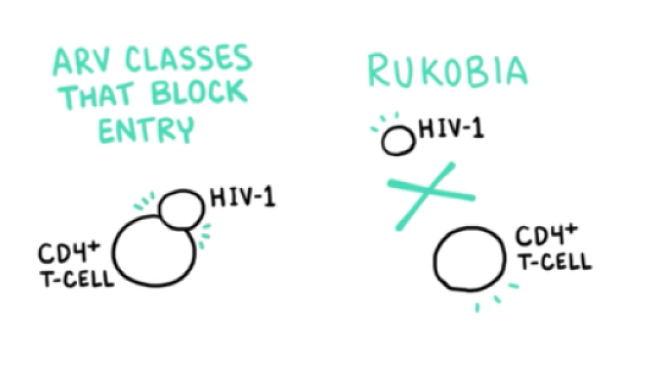
VO: Three antiretrovirals, that block entry...
SUPERS: HIV-1 viral envelope; CD4+ T-cell surface; gp120-CD4 attachment; ibalizumab.
VIDEO: The left of screen shows gp120 trimer and where ibalizumab acts following attachment with CD4 target. New supers appear one at a time.
VO: ... act following the attachment of the gp120 viral envelope protein...
SUPERS: HIV-1 viral envelope; CD4+ T-cell surface; co-receptor binding; maraviroc.
VIDEO: The ibalizumab screen fades out, and the middle of the screen animates in to show where maraviroc acts following attachment of gp120 trimer with the CD4 target. New supers appear one at a time.
VO: ... with the CD4 target. In comparison...
SUPERS: HIV-1 viral envelope; CD4+ T-cell surface; membrane fusion; enfuvirtide.
VIDEO: The maraviroc screen fades out, and the right of the screen animates in to show where enfuvirtide acts following attachment of the gp120 trimer with the CD4 target. New supers appear one at a time.
VO: ...blocking the attachment of gp120 prevents the earliest stage of HIV-1 and CD4+ T-cell engagement.
SUPERS: CD4+ T-cell; HIV-1; Lymph Node; RUKOBIA (fostemsavir).
VIDEO: Supers from Frame 8 fade out and animation transitions to cellular space showing CD4 cells and HIV-1 virions. New supers fade in. RUKOBIA (fostemsavir) approaches the HIV-1 surface.
VO: A first-in-class treatment with a novel mechanism of action, RUKOBIA is the first FDA-approved HIV-1 attachment inhibitor.
SUPERS: RUKOBIA (fostemsavir).
VIDEO: RUKOBIA (fostemsavir) molecule with HIV-1 surface and trimer in background.
VO: RUKOBIA prevents HIV-1 from interacting with host immune cells—leaving CD4+ T-cells untouched.
SUPERS: RUKOBIA (fostemsavir).
VIDEO: RUKOBIA (fostemsavir) molecule moves toward the HIV-1 trimer.
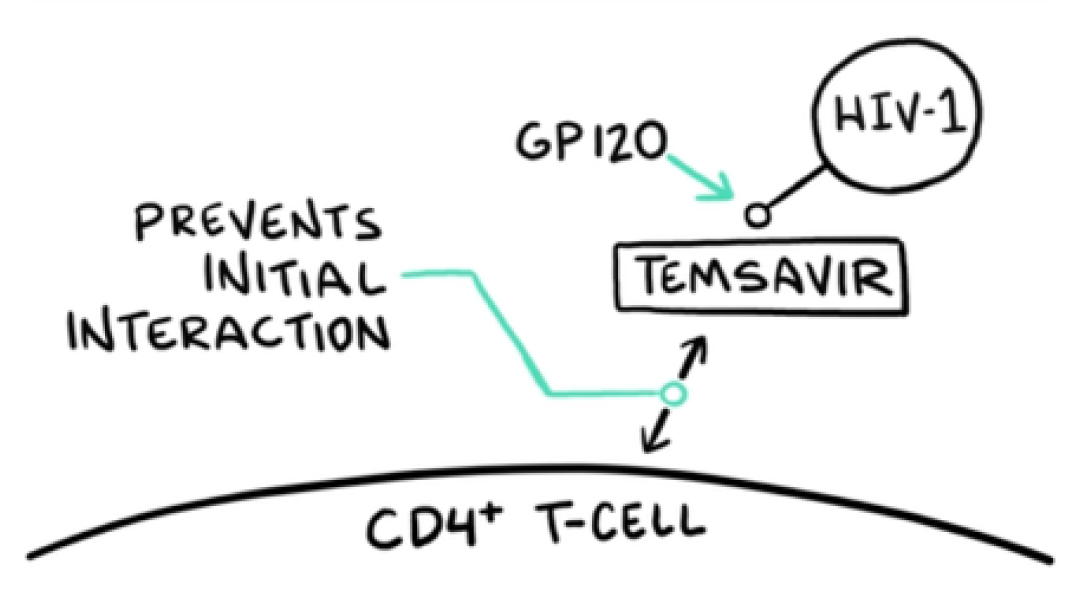
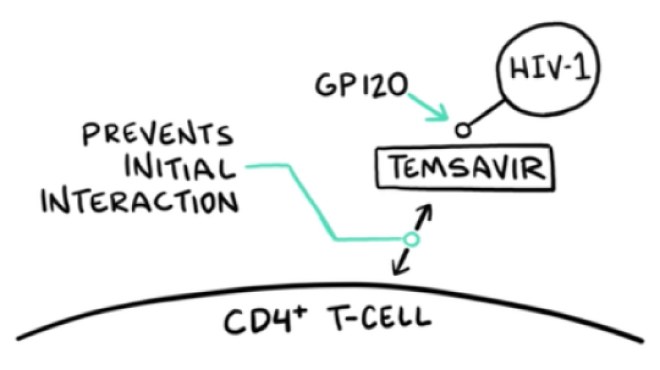
VO: Temsavir, the active moiety of fostemsavir, binds to the viral envelope protein gp120 to prevent initial HIV-1 attachment to CD4+ T-cells, the first step of HIV attachment and infection.
SUPERS: HIV-1 viral membrane; gp120; Temsavir, active moiety of RUKOBIA (fostemsavir).
VIDEO: The RUKOBIA (fostemsavir) molecule continues to move closer to the gp120 in the trimer. New supers fade in.
VO: Temsavir attaches directly to gp120 and selectively inhibits the interaction between the virus and cellular CD4 receptors, thereby preventing attachment and all post-attachment processes. The activity of temsavir was not affected by viral strain or tropism.
VIDEO: RUKOBIA (fostemsavir) molecule binds to gp120 in the trimer, locking it in the closed formation.
VO: The attachment of temsavir locks the gp120/gp41 trimer in a closed state to prevent binding to CD4 receptors.
SUPERS: Closed state.
VIDEO: With the trimer locked in the closed formation with RUKOBIA, HIV-1 cannot bind to the CD4 receptor.
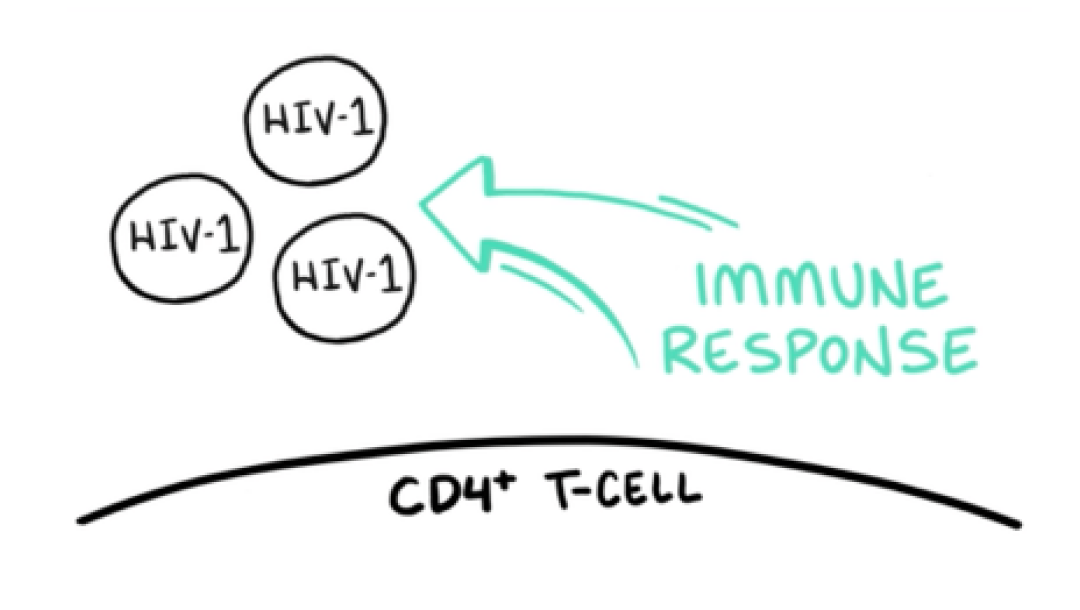
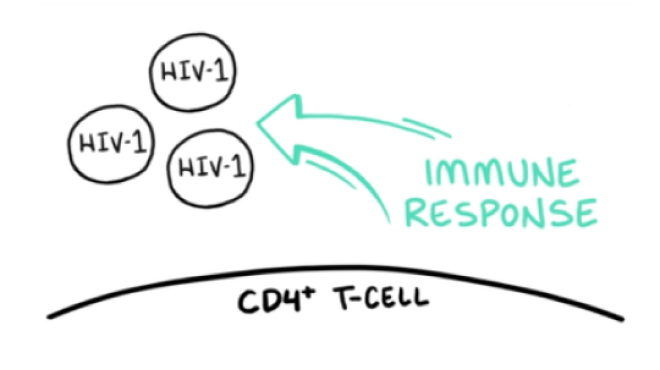
VO: With the HIV-1 virions unable to attach to CD4+ T-cells, they are a target for elimination by the body’s immune response.
SUPERS: Macrophage; Phagocytosis; Lymph node; HIV-1.
VIDEO: Transition into the cellular space with HIV-1 unable to bind with CD4 cells. New supers fade in.
VO: Important Safety Information, continued. Warnings and precautions. Immune Reconstitution Syndrome and autoimmune disorders have been reported with the use of RUKOBIA. QTc prolongation has occurred with higher than recommended dosages of RUKOBIA. Use RUKOBIA with caution in patients with a history of QTc interval prolongation or cardiac disease, or who are taking drugs with a known risk of Torsade de Pointes. Elderly patients may be more susceptible. Monitoring of liver chemistries is recommended in patients with hepatitis B and/or C co-infection. Diligence should be applied when starting RUKOBIA in patients receiving hepatitis B therapy. Adverse reactions or loss of virologic response due to drug interactions may occur when using RUKOBIA with other drugs.
VIDEO: Important Safety Information fades in and begins scrolling, pace keeping in time with VO.
VO: Adverse reactions. The most common adverse reaction reported with RUKOBIA was nausea. Drug interactions. See the full Prescribing Information for RUKOBIA for all significant drug interactions. Temsavir may increase plasma concentrations of grazoprevir and voxilaprevir. Use an alternative hepatitis C virus regimen, if possible. Use the lowest possible starting dose for statins with careful monitoring. Patients receiving RUKOBIA should not take estrogen-based therapies that contain more than 30 micrograms per day of ethinyl estradiol. Use RUKOBIA with caution in patients with additional risk factors for thromboembolic events.
VIDEO: Important Safety Information continues scrolling.
VO: Use in specific populations. There are insufficient data on the use of RUKOBIA during pregnancy to assess drug-associated risks. Breastfeeding is not recommended in patients with HIV-1. Please use the link on this page to view the full Prescribing Information for RUKOBIA.
VIDEO: Important Safety Information finishes scrolling and fades out.
VO: N/A
VIDEO: References fade in.
VO: N/A
SUPERS: Trademarks are owned by or licensed to the ViiV Healthcare group of companies. ©2023 ViiV Healthcare or licensor. FSTVID230005 October 2023 Produced in USA.
VIDEO: ViiV and RUKOBIA logos fade in with trademark line.
Temsavir, the active moiety of RUKOBIA, attaches directly to gp120 on the surface of HIV-1 virions, near the CD4+ attachment site. The attachment of temsavir locks gp120 into a closed formation that prevents the initial interaction between the virus and host immune cells.1,2
This action prevents the first step of viral entry.3
VO: RUKOBIA Educational Series
Visual: RUKOBIA EDUCATIONAL SERIES and logos appear on screen.
VO: Mechanism of action. Indication. RUKOBIA, used with other antiretrovirals, is indicated for heavily treatment-experienced adults with multidrug-resistant HIV-1 who are failing their current ARV regimen. Important Safety Information. Contraindications. Do not use in patients with previous hypersensitivity to fostemsavir or any of the components of RUKOBIA. Do not use RUKOBIA in patients receiving strong cytochrome P450 (CYP)3A inducers, including those listed here. Please see additional Important Safety Information for RUKOBIA at the end of the video. Please click the link on this page to view the full Prescribing Information for RUKOBIA.
Visual: MECHANISM OF ACTION appears on the screen and then fades out, followed by the fading in of the RUKOBIA indication, contraindications, redirect to the additional Important Safety Information at the end of the video, and link to the full Prescribing Information. The length of time supers appear on screen aligns with VO narration.
VO: First-in-class RUKOBIA
Visual: FIRST-IN-CLASS RUKOBIA is written on screen.
VO: is the first FDA-approved HIV-1 attachment inhibitor.
Visual: FIRST FDA-APPROVED HIV-1 ATTACHMENT INHIBITOR is written.
VO: Unlike previously approved classes that act by blocking entry following attachment of the glycoprotein 120, or gp120, viral envelope protein,
Visual: ARV CLASSES THAT BLOCK ENTRY, HIV-1, and CD4+ T-CELL are drawn.
VO: RUKOBIA acts on the HIV-1 virion before attachment of gp120 to prevent the earliest stage of HIV-1 and CD4+ T-cell engagement.
Visual: RUKOBIA, HIV-1, and CD4+ T-CELL are added to complete the frame.
VO: Temsavir, the active moiety of RUKOBIA, attaches to gp120 on the surface of HIV-1 virions, near the CD4 attachment site.
Visual: HIV-1, GP120, and TEMSAVIR are drawn.
VO: The attachment of temsavir locks gp120 into a closed formation that prevents the initial interaction between the virus and host immune cells, leaving CD4+ T-cells untouched.
Visual: PREVENTS INITIAL INTERACTION arm and CD4+ T-CELL membrane are drawn on screen.
VO: With the HIV-1 virions unable to attach to CD4+ T-cells, they are a target for elimination by the body’s immune response.
Visual: The HIV-1 virions and CD4+ T-cell are drawn. The arrow and IMMUNE RESPONSE are added to the frame.
VO: Important Safety Information, continued. Warnings and Precautions. Immune Reconstitution Syndrome and autoimmune disorders have been reported with the use of RUKOBIA. QTc prolongation has occurred with higher than recommended dosages of RUKOBIA. Use RUKOBIA with caution in patients with a history of QTc interval prolongation or cardiac disease, or who are taking drugs with a known risk of Torsade de Pointes. Elderly patients may be more susceptible. Monitoring of liver chemistries is recommended in patients with hepatitis B and/or C co-infection. Diligence should be applied when starting RUKOBIA in patients receiving hepatitis B therapy. Adverse reactions or loss of virologic response due to drug interactions may occur when using RUKOBIA with other drugs. Adverse Reactions. The most common adverse reaction reported with RUKOBIA was nausea.
Visual: Important Safety Information scrolls on screen.
VO: Drug Interactions. See the full Prescribing Information for RUKOBIA for all significant drug interactions. Temsavir may increase plasma concentrations of grazoprevir and voxilaprevir. Use an alternative hepatitis C virus regimen, if possible. Use the lowest possible starting dose for statins with careful monitoring. Patients receiving RUKOBIA should not take estrogen-based therapies that contain more than 30 micrograms per day of ethinyl estradiol. Use RUKOBIA with caution in patients with additional risk factors for thromboembolic events. Use in Specific Populations. There are insufficient data on the use of RUKOBIA during pregnancy to assess drug-associated risks. Breastfeeding is not recommended in patients with HIV-1. Please click the link on this page to view the full Prescribing Information for RUKOBIA.
Visual: Important Safety Information scrolls on screen.
REFERENCES:
- Thompson M, Lalezari JP, Kaplan R, et al. Safety and efficacy of the HIV-1 attachment inhibitor prodrug fostemsavir in antiretroviral-experienced subjects: week 48 analysis of AI438011, a Phase IIb, randomized controlled trial. Antivir Ther. 2017;22(3):215-223.
- FDA Approval of HIV Medicines. AIDSinfo website. https://aidsinfo.nih.gov/understanding-hiv-aids/infographics/25/fda-approval-of-hiv-medicines. Updated April 30, 2019. Accessed May 9, 2020.
- Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in adults and adolescents with HIV. Department of Health and Human Services. https://aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf. Updated December 18, 2019. Accessed May 7, 2020.
- Cahn P, Fink V, Patterson P. Fostemsavir: a new CD4 attachment inhibitor. Curr Opin HIV AIDS. 2018;13(4):341-345.
See how RUKOBIA prevents HIV-1 from interacting with host immune cells
References:
- Ackerman P, Thompson M, Molina JM, et al. Long-term efficacy and safety of fostemsavir among subgroups of heavily treatment-experienced adults with HIV-1 AIDS 2021;35(7):1061-1072.
- Thompson M, Lalezari JP, Kaplan R, et al. Safety and efficacy of the HIV-1 attachment inhibitor prodrug fostemsavir in antiretroviral-experienced subjects: week 48 analysis of AI438011, a Phase IIb, randomized controlled trial. Antivir Ther. 2017;22(3):215-223.
- Cahn P, Fink V, Patterson P. Fostemsavir: a new CD4 attachment inhibitor. Curr Opin HIV AIDS. 2018;13(4):341-345.